Summary
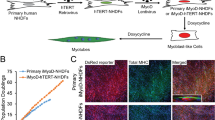
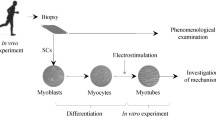
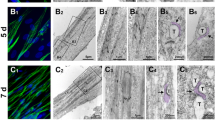
Our purpose was to engineer three-dimensional skeletal muscle tissue constructs from primary cultures of adult rat myogenic precursor cells, and to measure their excitability and isometric contractile properties. The constructs, termed myooids, were muscle-like in appearance, excitability, and contractile function. The myooids were 12 mm long and ranged in diameter from 0.1 to 1 mm. The myooids were engineered with synthetic tendons at each end to permit the measurement of isometric contractile properties. Within each myooid the myotubes and fibroblasts were supported by an extracellular matrix generated by the cells themselves, and did not require a preexisting scaffold to define the size, shape, and general mechanical properties of the resulting structure. Once formed, the myooids contracted spontaneously at approximately 1 Hz, with peak-to-peak force amplitudes ranging from 3 to 30 μN. When stimulated electrically the myooids contracted to produce force. The myooids (n=14) had the following mean values: diameter of 0.49 mm, rheobase of 1.0 V/mm, chronaxie of 0.45 ms, twitch force of 215 μN, maximum isometric force of 440 μN, resting baseline force of 181 μN, and specific force of 2.9kN/m2. The mean specific force was approximately 1% of the specific force generated by control adult rat muscle. Based on the functional data, the myotubes in the myooids appear to remain arrested in an early developmental state due to the absence of signals to promote expression of adult myosin isoforms.
Similar content being viewed by others
References
Brooke, M. H.; Kaiser, K. K. Muscle fiber types: how many and what kind? Arch. Neurol. 23:369–379; 1970.
Close, R. Dynamic properties of fast and slow skeletal muscles of the rat during development. J. Physiol. 173:74–95; 1964.
Delvoye, P.; Wiliquet, P.; Leveque, J.; Nusgens, B. V.; Lapiere, C. M. Measurement of mechanical forces generated by skin fibroblasts embedded in a three-dimensional collagen gel. J. Investig. Dermatol. 97: 898–902; 1991.
Faulkner, J. A.; Brooks, S. V.; Dennis, R. G. Measurement of recovery of function following whole muscle transfer, myoblast transfer, and gene therapy. In: Morgan, J. R.; Yarmush, M. L., ed., Methods in tissue engineering, vol. 18. Tissue engineering methods and protocols. Totowa, NJ: Humana Press; 1997:155–172.
Gordon, A. M.; Huxley, A. F.; Julian, F. J. The variation in isometric tension with sarcomere length in vertebrate muscle fibres. J. Physiol. (London) 184(1):170–192; 1966.
Guth, L; Samaha, F. J. Erroneous interpretations which may result from application of the “myofibrillar ATPase” histochemical procedure to developing muscle. Exp. Neurol. 34:465–475; 1972.
Hatfaludy, S.; Shansky, J.; Vandenburgh, H. H. Metabolic alterations induced in cultured skeletal muscle by stretch-relaxation activity. Am. J. Physiol. 256(1 Pt 1):C175-C181; 1989.
Lewis, M. R. Rhythmical contraction of the skeletal muscle tissue observed in tissue cultures. Am. J. Physiol. 38:153–161; 1915.
Minns, H. G. A voltage-controlled force generator for calibrating sensitive transducers. J. Appl. Physiol. 30(6):895–896; 1971.
Okano, T.; Matsuda, T. Muscular tissue engineering: capillary-incorporated hybrid muscular tissues in vivo tissue culture. Cell Transplant 7(5):435–442; 1998.
Okano, T.; Satoh, S.; Oka, T.; Matsuda, T. Tissue engineering of skeletal muscle: highly dense, highly oriented hybrid muscular tissues biomimicking native tissues. ASAIO J. 43:M749-M753; 1997.
Perrone, C. E.; Fenwick-Smith, D.; Vandenburgh, H. H. Collagen and stretch modulate autocrine secretion of insulin-like growth factor-1 and insulin-like growth factor binding proteins from differentiated skeletal muscle cells. J. Biol. Chem. 270(5):2099–2106; 1995.
Schultz, E.; McCormick, K. M. Skeletal muscle satellite cells. Rev. Physiol. Biochem. Pharmacol. 123:213–257; 1994.
Shansky, J.; Chromiak, J.; Del Tatto, M.; Vandenburgh, H. A simplified method for tissue engineering skeletal muscle organoids in vitro. In Vitro Cell. Dev. Biol. Anim. 33:659–661; 1997.
Strohman, R. C.; Bayne, E.; Spector, D.; Obinata, T.; Micou-Eastwood, J.: Maniotis, A. Myogenesis and histogenesis of skeletal muscle on flexible membranes in vitro. In Vitro Cell. Dev. Biol. 26(2):201–208; 1990.
Swasdison, S.; Mayne, R. In vitro attachment of skeletal muscle fibers to a collagen gel duplicates the structure of the myotendinous junction. Exp. Cell. Res. 193:227–231; 1991.
Swasdison, S.; Mayne, R. Formation of highly organized skeletal muscle fibers in vitro: comparison with muscle development in vivo. J. Cell. Sci. 102:643–652; 1992.
Vandenburgh, H. H. Dynamic mechanical orientation of skeletal myofibers in vitro. Dev. Biol. 93(2):438–443;, 1982.
Vandenburgh, H. H. A computerized mechanical cell stimulator for tissue culture: effects on skeletal muscle organogenesis. In Vitro Cell. Dev. Biol. 24(7):609–619; 1988.
Vandenburgh, H. H.; Karlisch, P. Longitudinal growth of skeletal myotubes in vitro in a new horizontal mechanical cell stimulator. In Vitro Cell. Dev. Biol. 25(7):607–616; 1989.
Vandenburgh, H. H. Mechanical forces and their second messengers in stimulating cell growth in vitro. Am. J. Physiol. 262(3 Pt 2):R350-R355; 1992.
Vandenburgh, H.; Del Tatto, M.; Shansky, J., et al. Tissue-engineered skeletal muscle organoids for reversible gene therapy. Hum. Gene. Ther. 7(17):2195–2200; 1996.
Vandenburgh, H. H.; Hatfaludy, S.; Karlisch, P.; Shansky, J. Skeletal muscle growth is stimulated by intermittent stretch-relaxation in tissue culture. Am. J. Physiol. 256(3 Pt 1):C674-C682; 1989.
Vandenburgh, H. H.; Hatfaludy, S.; Karlisch, P.; Shansky, J. Mechanically induced alterations in cultured skeletal muscle growth. J. Biomech. 24(Suppl. 1):91–99; 1991a.
Vandenburgh, H. H.; Hatfaludy, S.; Sohar, I.; Shansky, J. Stretch-induced prostaglandins and protein turnover in cultured skeletal muscle. Am. J. Physiol. 259(2 Pt. 1):C232-C240; 1990.
Vandenburgh, H.; Kaufman, S. In vitro model for stretch-induced hypertrophy of skeletal muscle. Science 203(4377):265–268; 1979.
Vandenburgh, H. H.; Swasdison, S.; Karlisch, P. Computer-aided mechanogenesis of skeletal muscle organs from single cells in vitro. FASEB J. 5(13):2860–2867; 1991b.
van Wachem, P. B.; van Luyn, M. J. A.; Ponte da Costa, M. L. Myoblast seeding in a collagen matrix evaluated in vitro. J. Biomed. Mat. Res. 30:353–360; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dennis, R.G., Kosnik, P.E. Excitability and isometric contractile properties of mammalian skeletal muscle constructs engineered in vitro. In Vitro Cell.Dev.Biol.-Animal 36, 327–335 (2000). https://doi.org/10.1290/1071-2690(2000)036<0327:EAICPO>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2000)036<0327:EAICPO>2.0.CO;2